EMBRYOPSIDA Pirani & Prado
Gametophyte dominant, independent, multicellular, initially ±globular, not motile, branched; showing gravitropism; glycolate oxidase +, glycolate metabolism in leaf peroxisomes [glyoxysomes], acquisition of phenylalanine lysase* [PAL], flavonoid synthesis*, microbial terpene synthase-like genes +, triterpenoids produced by CYP716 enzymes, CYP73 and phenylpropanoid metabolism [development of phenolic network], xyloglucans in primary cell wall, side chains charged; plant poikilohydrous [protoplasm dessication tolerant], ectohydrous [free water outside plant physiologically important]; thalloid, leafy, with single-celled apical meristem, tissues little differentiated, rhizoids +, unicellular; chloroplasts several per cell, pyrenoids 0; centrioles/centrosomes in vegetative cells 0, microtubules with γ-tubulin along their lengths [?here], interphase microtubules form hoop-like system; metaphase spindle anastral, predictive preprophase band + [with microtubules and F-actin; where new cell wall will form], phragmoplast + [cell wall deposition centrifugal, from around the anaphase spindle], plasmodesmata +; antheridia and archegonia +, jacketed*, surficial; blepharoplast +, centrioles develop de novo, bicentriole pair coaxial, separate at midpoint, centrioles rotate, associated with basal bodies of cilia, multilayered structure + [4 layers: L1, L4, tubules; L2, L3, short vertical lamellae] (0), spline + [tubules from L1 encircling spermatid], basal body 200-250 nm long, associated with amorphous electron-dense material, microtubules in basal end lacking symmetry, stellate array of filaments in transition zone extended, axonemal cap 0 [microtubules disorganized at apex of cilium]; male gametes [spermatozoids] with a left-handed coil, cilia 2, lateral, asymmetrical; oogamy; sporophyte +*, multicellular, growth 3-dimensional*, cuticle +*, plane of first cell division transverse [with respect to long axis of archegonium/embryo sac], sporangium and upper part of seta developing from epibasal cell [towards the archegonial neck, exoscopic], with at least transient apical cell [?level], initially surrounded by and dependent on gametophyte, placental transfer cells +, in both sporophyte and gametophyte, wall ingrowths develop early; suspensor/foot +, cells at foot tip somewhat haustorial; sporangium +, single, terminal, dehiscence longitudinal; meiosis sporic, monoplastidic, MTOC [= MicroTubule Organizing Centre] associated with plastid, sporocytes 4-lobed, cytokinesis simultaneous, preceding nuclear division, quadripolar microtubule system +; wall development both centripetal and centrifugal, 1000 spores/sporangium, sporopollenin in the spore wall* laid down in association with trilamellar layers [white-line centred lamellae; tripartite lamellae]; plastid transmission maternal; nuclear genome [1C] <1.4 pg, main telomere sequence motif TTTAGGG, KNOX1 and KNOX2 [duplication] and LEAFY genes present, ethylene involved in cell elongation; chloroplast genome with close association between trnLUAA and trnFGAA genes [precursors for starch synthesis], tufA, minD, minE genes moved to nucleus; mitochondrial trnS(gcu) and trnN(guu) genes +.
Many of the bolded characters in the characterization above are apomorphies of more or less inclusive clades of streptophytes along the lineage leading to the embryophytes, not apomorphies of crown-group embryophytes per se.
All groups below are crown groups, nearly all are extant. Characters mentioned are those of the immediate common ancestor of the group, [] contains explanatory material, () features common in clade, exact status unclear.
POLYSPORANGIOPHYTA†
Sporophyte well developed, branched, branching dichotomous, potentially indeterminate; hydroids +; stomata on stem; sporangia several, terminal; spore walls not multilamellate [?here].
II. TRACHEOPHYTA / VASCULAR PLANTS
Sporophyte long lived, cells polyplastidic, photosynthetic red light response, stomata open in response to blue light; plant homoiohydrous [water content of protoplasm relatively stable]; control of leaf hydration passive; plant endohydrous [physiologically important free water inside plant]; PIN[auxin efflux facilitators]-mediated polar auxin transport; (condensed or nonhydrolyzable tannins/proanthocyanidins +); borate cross-linked rhamnogalactan II, xyloglucans with side chains uncharged [?level], in secondary walls of vascular and mechanical tissue; lignins +; roots +, often ≤1 mm across, root hairs and root cap +; stem apex multicellular [several apical initials, no tunica], with cytohistochemical zonation, plasmodesmata formation based on cell lineage; vascular development acropetal, tracheids +, in both protoxylem and metaxylem, G- and S-types; sieve cells + [nucleus degenerating]; endodermis +; stomata numerous, involved in gas exchange; leaves +, vascularized, spirally arranged, blades with mean venation density ca 1.8 mm/mm2 [to 5 mm/mm2], all epidermal cells with chloroplasts; sporangia in strobili, sporangia adaxial, columella 0; tapetum glandular; sporophyte-gametophyte junction lacking dead gametophytic cells, mucilage, ?position of transfer cells; MTOCs not associated with plastids, basal body 350-550 nm long, stellate array in transition region initially joining microtubule triplets; archegonia embedded/sunken [only neck protruding]; embryo suspensor +, shoot apex developing away from micropyle/archegonial neck [from hypobasal cell, endoscopic], root lateral with respect to the longitudinal axis of the embryo [plant homorhizic].
[MONILOPHYTA + LIGNOPHYTA]Sporophyte growth ± monopodial, branching spiral; roots endomycorrhizal [with Glomeromycota], lateral roots +, endogenous; G-type tracheids +, with scalariform-bordered pits; leaves with apical/marginal growth, venation development basipetal, growth determinate; sporangium dehiscence by a single longitudinal slit; cells polyplastidic, MTOCs diffuse, perinuclear, migratory; blepharoplasts +, paired, with electron-dense material, centrioles on periphery, male gametes multiciliate; nuclear genome [1C] 7.6-10 pg [mode]; chloroplast long single copy ca 30kb inversion [from psbM to ycf2]; mitochondrion with loss of 4 genes, absence of numerous group II introns; LITTLE ZIPPER proteins.
LIGNOPHYTA†
Sporophyte woody; stem branching axillary, buds exogenous; lateral root origin from the pericycle; cork cambium + [producing cork abaxially], vascular cambium bifacial [producing phloem abaxially and xylem adaxially].
SEED PLANTS† / SPERMATOPHYTA†
Growth of plant bipolar [plumule/stem and radicle/root independent, roots positively geotropic]; plants heterosporous; megasporangium surrounded by cupule [i.e. = unitegmic ovule, cupule = integument]; pollen lands on ovule; megaspore germination endosporic, female gametophyte initially retained on the plant, free-nuclear/syncytial to start with, walls then coming to surround the individual nuclei, process proceeding centripetally.
EXTANT SEED PLANTS
Plant evergreen; nicotinic acid metabolised to trigonelline, (cyanogenesis via tyrosine pathway); microbial terpene synthase-like genes 0; primary cell walls rich in xyloglucans and/or glucomannans, 25-30% pectin [Type I walls]; lignin chains started by monolignol dimerization [resinols common], particularly with guaiacyl and p-hydroxyphenyl [G + H] units [sinapyl units uncommon, no Maüle reaction]; roots often ≥1 mm across, stele diarch to pentarch, xylem and phloem originating on alternating radii, cork cambium deep seated, gravitropism response fast; stem apical meristem complex [with quiescent centre, etc.], plasmodesma density in SAM 1.6-6.2[mean]/μm2 [interface-specific plasmodesmatal network]; eustele +, protoxylem endarch, endodermis 0; wood homoxylous, tracheids and rays alone, tracheid/tracheid pits circular, bordered; mature sieve tube/cell lacking functioning nucleus, sieve tube plastids with starch grains; phloem fibres +; cork cambium superficial; leaf nodes 1:1, a single trace leaving the vascular sympodium; leaf vascular bundles amphicribral; guard cells the only epidermal cells with chloroplasts, stomatal pore with active opening in response to leaf hydration, control by abscisic acid, metabolic regulation of water use efficiency, etc.; branching by axillary buds, exogenous; prophylls two, lateral; leaves with petiole and lamina, development basipetal, lamina simple; sporangia borne on sporophylls; spores not dormant; microsporophylls aggregated in indeterminate cones/strobili; grains monosulcate, aperture in ana- position [distal], primexine + [involved in exine pattern formation with deposition of sporopollenin from tapetum there], exine and intine homogeneous, exine alveolar/honeycomb; ovules with parietal tissue [= crassinucellate], megaspore tetrad linear, functional megaspore single, chalazal, sporopollenin 0; gametophyte ± wholly dependent on sporophyte, development initially endosporic [apical cell 0, rhizoids 0, etc.]; male gametophyte with tube developing from distal end of grain, male gametes two, developing after pollination, with cell walls; embryo cellular ab initio, suspensor short-minute, embryonic axis straight [shoot and root at opposite ends], primary root/radicle produces taproot [= allorhizic], cotyledons 2; embryo ± dormant; chloroplast ycf2 gene in inverted repeat, trans splicing of five mitochondrial group II introns, rpl6 gene absent; ??whole nuclear genome duplication [ζ/zeta duplication event], 2C genome size (0.71-)1.99(-5.49) pg, two copies of LEAFY gene, PHY gene duplications [three - [BP [A/N + C/O]] - copies], 5.8S and 5S rDNA in separate clusters.
bipolar [plumule/stem and radicle/root independent, roots positively geotropic];EXTANT GYMNOSPERMS / ACROGYMNOSPERMAE
Biflavonoids +; ferulic acid ester-linked to primary unlignified cell walls, silica usu. low; root apical meristem organization?, protophloem not producing sieve tubes, with secretory cells, sieve area of sieve tube with small pores generally less than 0.8 µm across that have cytoplasm and E.R., joining to form a median cavity in the region of the middle lamella, Strasburger/albuminous cells associated with sieve tubes [the two not derived from the same immediate mother cell], phloem fibres +; sclereids +, ± tracheidal transfusion tissue +, rays uniseriate [?here]; stomatal poles raised above pore, no outer stomatal ledges or vestibule, epidermis lignified; cuticle waxes as tubules, nonacosan-10-ol predominates, n-alkyl lipids scanty; buds perulate/with cataphylls; leaves simple, lamina 1-veined, development marginal; plants dioecious; parts of strobili spirally arranged; microsporangia abaxial, dehiscing by the action of the epidermis [= exothecium]; pollen saccate, tectate, endexine lamellate at maturity all around grain, esp. intine with callose; ovules aggregated into compound strobili, erect, pollen chamber formed by breakdown of nucellar cells, nucellus massive; ovules increasing considerably in size between pollination and fertilization, but aborting unless pollination occurs; ovule with pollination droplet, catches pollen; male gametophyte: two prothallial cells + tube cell + antheridial cell, the latter → sterile cell + 2 gametes; pollen germinates on ovule, usu. takes two or more days, tube with wall of pectose + cellulose microfibrils, branched, growing at up to 10(-20) µm/h-1, haustorial, breaks down sporophytic cells; male gametes released by breakdown of pollen grain wall, with >1000 cilia, basal body 800-900 nm long, spline hundreds of tubules wide, chromatin not condensed; fertilization 7 days to 12 months or more after pollination, to ca 2 mm from ovule surface to egg; seeds "large" [ca 8 mm3], but not much bigger than ovule, with morphological dormancy; testa mainly of coloured sarcoexotesta, scleromesotesta, and ± degenerating endotesta; first zygotic nuclear division with chromosomes of male and female gametes lining up on separate but parallel spindles, embryogenesis initially nuclear, embryo ± chlorophyllous; gametophyte persists in seed; plastid and mitochondrial transmission paternal; genome size [1C] 10< pg [1 pg = 109 base pairs]/(2201-)17947(-35208) Mb; two copies of LEAFY gene [LEAFY, NEEDLY] and three of the PHY gene, [PHYP [PHYN + PHYO]]; plastome IR expanded, with duplicated ribosomal RNA operons; chondrome with second intron in the rps3 gene [group II, rps3i2].
[CUPRESSALES [GNETALES + PINALES]] / PINOPHYTA / CONIFEROPHYTA / CONIFERAE / PINOPSIDA
Tree, branched, evergreen; βaryl ether concentration in lignin lower, biphenyls higher; compression wood + [reaction wood - much-thickened/lignified fibres on abaxial side of branch-stem junction]; wood pycnoxylic, tracheids with torus:margo pits + [tracheid side walls]; phloem with polyphenol-containing parenchyma (PP) cells, resin canals/cells in phloem and/or xylem +/0; lignins with guaiacyl units (G-lignin) [lacking syringaldehyde, Mäule reaction negative]; cork cambium ± deep seated; bordered pits on tracheids round, opposite; nodes 1:1; axillary buds + (0); leaves with single vein, fasciculate or not, needle-like or flattened; plants monoecious; microsporangiophore/filament simple, hyposporangiate; dehiscing by the action of the hypodermis [endothecium]; pollen saccate, exine thick [³2 µm thick], granular; ovulate strobilus compound, erect, ovuliferous scales flattened, ± united with bract scales [= ovules borne on predeveloped axillary structures]; ovules with pollination drop, lacking pollen chamber, inverted [micropyle facing cone axis at pollination and seed dispersal]; pollen buoyant, not wettable, pollen tube unbranched, not haustorial, growing towards ovule, development intercellular, wall with cellulose and arabinogalactan proteins; gametes non-motile, lacking walls, siphonogamy [released from distal end of tube directly to the egg cell]; female gametophyte lacking chlorophyll; seed cone components sclerified, seed dispersed with part of supporting structure, seed coat dry, not vascularized; embryo initially with 2 to 4 free-nuclear divisions, elongated suspensor cells, embryonal cells basal, polyembryony +; one duplication in the PHYP gene line; germination phanerocotylar, epigeal, (seedlings green in the dark).
[GNETALES + PINALES] (if the clade exists): phloem fibres 0; microsporangiophore/filament simple with terminal microsporangia; microsporangia abaxial, dehiscing by the action of the hypodermis [endothecium]; lhcb 3 and 6 genes not functional/lost [light harvesting genes]; chloroplast transmission maternal, ndh genes lost/pseudogenized, rpl16 gene lost.
PINALES Link / Conifers I - Main Tree.
Just the one family, 11 genera, 231 species.
Phylogeny. For information on the other major seed plant groups, see also angiosperms, Cupressales, Cycadales, Ginkgoales and Gnetales, and for the relationships of Pinales/Pinaceae, see elsewhere.
Synonymy: Abietales Link - Pinidae Cronquist, Takhtajan, & Zimmermann - Pinopsida Burnett - Pinophytina Reveal
PINACEAE F. Rudolphi - Back to Pinales
Plant (deciduous), ectomycorrhizal; specialized resin diterpenes [e.g. with abietane/pimarane skeletons], biflavonoids 0, (2-6-piperidine alkaloids +); root hairs +; reaction wood on lower side of stem, xylem resin canals +, inducible/traumatic, also constitutive; vessels 0; phloem with constitutive resiniferous structures, sieve cells with nacreous walls, sieve tube plastids also with protein fibres, crystals frequent in marginal and submarginal cells of ray parenchyma, horizontal walls of radial ray parenchyma cells distinctly pitted; annual rings of polyphenol parenchyma cells, sclereids scattered (in groups), calcium oxalate crystals large, intracellular; cortical resin canals +/0; wax deposits filling the stomatal chamber; axillary buds common, (short shoots, spur shoots +); lamina vascular bundle surrounded by sheath, adaxial palisade tissue +/0; 2 microsporangia/microsporophyll, sporangia superficial; pollen saccate, exine thin [2³ µm] except distally, alveolar/honeycomb; male gametophyte: pollen shed at 4-nucleate stage, 6-nucleate - 2 prothallial cells/tube cell/sterile cell/gametes; seed cones terminal (axillary), persistent, bract scale free from ovuliferous scale; ovules 2/ovuliferous scale, inverted, pollination droplet +; seed takes 2 (pollen tubes dormant) years to develop; early embryo: proembryo with 4 nuclei in initial phase of free nuclear divisions [= embryo tetrad], immediately afterwards with 4 tiers, the basal tier embryo proper, the next tier suspensor, then two more tiers; (bract scale deciduous); seeds dry, wing + [developing from adaxial tissues of the ovuliferous scale], terminal/(0); (integument with resin canals); simple (and cleavage, etc.) polyembryony +; polycotyly [cotyledons (2-)4-11(-20)]; n = x = 12 (22 - Pseudolarix), nuclear genome [1 C] 9-36 pg; PHYP gene duplicated, ndh complex 0, plastome IRb copy lost.
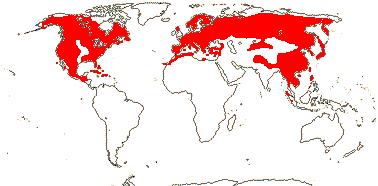
11 [list]/231. North Temperate (map: from Florin 1963; Farjon 1984, 1990a). [Photos - Collection.]
Age. Magallón et al. (2013) suggested an age of (161.2-)153.8-153.1(-150.1) Ma for crown-group Pinaceae, He et al. (2012) an age of around 237 Ma. Various divergence estimates were provided by Gernandt et al. (2008), e.g. dates of ca 184 Ma (Jurassic) or ca 136 Ma, about 198.4 Ma is the estimate in Y. Lu et al. (2014) while around 175 Ma is that in Leslie et al. (2012) similar to the estimate of ca 172 Ma in Ran et al. (2018a), not that different from the estimate of (226.6-)189.1(-155.2) Ma in Leslie et al. (2018); crown and stem ages of 100 and 263 Ma respectively were suggested by Quirk et al. (2012); see also Tedersoo and Brundrett (2017). Ca 48 Ma is the estimate in Evkaikina et al. (2017), (266.9-)206.3(-168) Ma in Ran et al. (2018b), 220-191.4 Ma in Semerikova et al. (2018), (233.4-)183.3(-150) Ma in Lutzoni et al. (2018) and 162-118 Ma in Herting et al. (2020).
Fossils assigned to Pinaceae have been dated to around 155 Ma (Rothwell et al. 2012; see also Miller 1999). Schizolepidopsis, from the Early Cretaceous, seems best assigned to stem-group Pinaceae (Andruchow-Colombo et al. 2023b).
Cognato and Grimaldi (2009) described a scolytine bark beetle = weevil, Microborus inertus, in ca 100 Ma amber from resin perhaps produced by Metasequoia from Myanmar, although Microborus probably infested angiosperms. Scolytines are diverse in Baltic amber ca 45 Ma produced by Pinites succinifera - linking phylogeny with these fossils may well be of interest.
1. Abietoideae Sweet —— Synonymy: Abietaceae Gray, Cedraceae Vest
Taproot with a single central resin canal; xylem with (constitutive axial canals)/inducible resin cavities/axial canals, phloem with constitutive cavities/radial canals; lamina (apically with 2 small teeth/lobes); two-year reproductive cycle ; (pollen not buoyant, wettable); ovule integument ca 13 cells across, parietal tissue 9-13 cells across; seed cones usu. erect, (not persistent); resin vesicles in seed coat; (n = 22 - Pseudolarix).
5/64. North Temperate to Boreal, mountains, Central America, North Africa.
Age. He et al. (2012) suggested an age of ca 200 Ma, Gernandt et al. (2008) ages of ca 172 or ca 109 Ma, Ran et al. (2018b) ages of (232.2-)170.2(-117.2) Ma, Lutzoni et al. (2018) ages of (233.4-)183.2(-150) Ma and Leslie et al. (2018) ages of (185.3-)141.0(-92.7) Ma for the beginning of Abietoideae diversification.
1A. Cedreae van Tieghem - Cedrus Trew
Short shoots +; vascular pits with scalloped torus margins; traumatic resin canals only, axial and radial; microsporopyll terminal; three-year reproductive cycle, fertilization in 3rd year, pollen grains rest for 7-8 months; male gametophyte c.f. Cephalotaxus; megaspore deep seated; cleavage polyembryony +; integument 4-6 cells across; seed dispersed as unit with all its supporting structures, integument multiplicative, outer 3-4 layers fleshy, become dry and papery, 4-5 layers stony cells, 6-7 inner layers of fleshy cells; embryo length of seed, cotyledons 8-14.
1/2-4. North Africa, Cyprus, Near East, Himalayas.
Age. Crown-group Cedrus is (14.0-)7.1(-1.7) Ma (Leslie et al. 2018).
[Abieteae + Pseudolariceae] : ?
1B. Abieteae Dumortier
Axillary buds 3 [= hastate - Ab.], (epicormic buds +); (pollination droplet 0 - Ab.); (seed dispersed as unit with all its supporting structures, cone axis persists - Abies); (germination hypogeal - Keteleeria).
2/51: Abies (48). North Temperate, to Central America, Taiwan, Laos and Vietnam.
Age. Crown-group Abieteae has been estimated to be (73.0-)58.1(-50.2) Ma (Leslie et al. 2018) or ca 67.7 Ma (Z.-R. Wei et al. 2025).
1C. Pseudolariceae L. Chu Li
(Pollen with circular inflated frill, exine spiny - Tsuga).
3/11: Tsuga (9). Temperate North America (Tsuga), East Asia (all three).
Age. Pseudolariceae are some (114.3-)83.0(-54.3) Ma (Leslie et al. 2018).
2. Pinoideae W. Hochstetter —— Synonymy: Piceaceae Gorozhankin
Branchlets ultimately abscise [cladoptosis] or not: taproot with resin canals associated with protoxylem poles; phloem with constitutive radial canals; xylem with constitutive network of resin canals, inducible axial canals, phloem with constitutive radial canals; leaves petiolate, lamina (apparently with two vascular bundles); integument ca 11 cells across; seed cones often pendulous; chromosomes large, 4.4-16.2 μm long; plastid transmission biparental, ndh genes 0 [Pinus]; (mitochondrial transmission maternal - Pinus).
5/167. North temperate to Boreal, mountains, to West Malesia.
Age. Approximate ages for crown-group Pinoideae are ca 168 or ca 133 Ma (Gernandt et al. 2008), ca 155 Ma (He et al. 2012), (203.7-)172.1(-145.7) Ma (Leslie et al. 2018) and (236.2-)185.4(-154-5) Ma (Ran et al. 2018b).
2A. Lariceae Rouy
(Plant deciduous - Larix; pollen not saccate, atectatey, exine granular; pollen not buoyant, pollination drop 0,exine shed during microgametophyte germination; (cones mature in one year - Larix), nuclear genome 10.9-14.7 Gb, (n = 13 - Pseudotsuga).
2/14: Larix (10). North Temperate.
Age. Crown Lariceae have been dated to (58.4-)47.0(-41.2) Ma (Leslie et al. 2018).
2B. Pineae Bluff & Fingerhuth
Crystals infrequent in marginal and submarginal cells of ray parenchyma; needles with 2 resin ducts; microsporophylls axillary/terminal; pollen chamber formed by breakdown of nucellar tissue [Pinus, Picea]; (male gametes with incomplete cell wall - Picea); (seed takes (1 year to develop - Picea/3 years to develop [pollen tube, then embryo dormant, fertilization in 3rd year] - Pinus); nuclear genome 12-17.6 Gb.
3/157: Pinus (118), Picea (38). North Temperate, to Central America, Sumatra and Java.
Age. The age of Pineae is some (147.0-)138.0(-133.2) Ma (Leslie et al. 2018).
Evolution: Divergence & Distribution. For more ages, see Leslie et al. (2018), and these authors also evaluate the rich fossil record which, however, shows only limited congruence with the ages they suggest. Cathaya, a monotypic genus now restricted to China, is known from Oligocene and later megafossils from Europe, while its distinctive pollen is known from various places in the northern hemisphere, including the Canadian Arctic, since the Cretaceous (Y.-S. Liu & Basinger 2000).
There may have been extinction in Pinaceae after initial diversification, generic-level crown-group diversification taking place within the last 50 Ma or so - and Pinaceae themselves have a stem ca 110 Ma (Leslie et al. 2018: no Gnetales). Similarly, despite the Triassic-Jurassic crown-group age for Pinaceae suggested by Semerikova et al. (2018), these authors thought that diversification began around the Palaeogene-Neogene transition, a mere ca 23 Ma, while Crisp and Cook (2011) estimated an age of around 27 Ma.
Pinus yorkshirensis, a cone associated with needles from Lower Cretaceous deposits 131-129 Ma, formed a polytomy with extant and some other fossil species of the genus in morphological analyses (Ryberg et al. 2012), no strong support being found for any Cretaceous fossils ascribed to the genus to nest within it (Klymiuk et al. 2011; Ryberg et al. 2011, and see Hilton et al. 2016 and references in Saladin et al. 2017). However, Gernandt et al. (2018b) found that P. yorkshirensis nested within crown-group Pinus, albeit with little support, as did a number of other fossils.
However, ages suggested for the diversification of Pinus in particular are all over the place. Thus stem ages for Pinus are around 95 or 73 Ma (B. Wang & Wang 2014), ca 126 Ma (He et al. 2012), ca 140 Ma (X.-Q. Wang et al. (2000), ca 123 or 155 Ma (Gernandt et al. 2008) or ca 180 Ma (Lockwood et al. 2013: Picea is sister). On the other hand, Naumann et al. (2013) date the Pinus/Picea split to around 33.1-29.3 Ma (angiosperms were the focus of this study). More: A stem-group age for Pinus of (132-)128(-124) Ma was suggested by Eckert and Hall (2006), while Crisp and Cook (2011) suggested that Pinus and Picea diverged around the K/C boundary ca 65 Ma. For other divergence times within Pinaceae, see also X.-Q. Wang et al. (2000), Lin et al. (2010), Saladin et al. (2017), Semerikova et al. (2018), and Ran et al. (2018b) and references.
J. W. Clark and Donoghue (2018) compared the morphospace occupied by Pinales, Cupressales, Gnetales and "Early Conifers", and they found that Pinales occupied a far greater morphospace than the other groups, indeed, even if those groups were combined. They linked this to a genome duplication that occurred very approximately 300-200 or 340-200 Ma (estimates from their Fig. 1 and Box 4 respectively) - but see Genes & Genomes below for conflicting ideas as to whether or not there has been a genome duplication shared by all Pinaceae.
X.-Q. Wang and Ran (2014) discuss the biogeography of the family. Pinus seems to have been a mid-latitude (30-50o N) plant in the Cretaceous, but in the warm Palaeocene and Eocene it retreated to higher latitudes, although also persisting near the equator. With the climatic deterioration of the Late Eocene-Oligocene, it moved back to mid latitudes while remaining at higher latitudes (Miller 1993), so Pinaceae could be quite common in high latitude Canadian Eocene floras (N. McIver & Basinger 1999). Le Page (2003; see also X.-Q. Wang et al. 2000) thought that there was an episode of diversification in Pinaceae in the Palaeocene. Sundaman et al. (2019) looked at biodiversity hotspots in conifers in general, noting that 80% or more of the species in them are not restricted to these hotspots, however, in the West Mexican hotspot there has been a fair amount of diversification of Pinus in particular, while Pinaceae are also important components of the Cascades-Sierra Nevada, Chinese and Japanese hotspots.
Extinction rates in conifers in general may have increased during the period 110-100 Ma, i.e. near the beginning of the Cretaceous Terrestrial Revolution, and this is perhaps the result of competition between conifers and angiosperms. Extinction rates have remained high and speciation rates low; the former may be due to more rapid growth, animal pollination, etc., in angiosperms and the latter in part to long generation time, large genome size, etc. (Condamine et al. 2020 and references). This conifer decline is indeed of long standing, furthermore, the rate of conifer decline may have increased as temperatures decreased in the Oligocene (Condamine et al. 2020), although W. T. Jin et al. (2021) show a steady, uninterrupted decline over the last 100 million years.
Turning to crown Pinus, estimates in Gernandt et al. (2008) are ca 87-72 Ma, and here and elsewhere its stem can be very long - in this case, ca 70 and 50 Ma respectively. Other estimates are much older, 165-148 Ma, or (144-)125, 112(-5) Ma (Saladin et al. 2017: another estimate, not favoured, ca 90 Ma) and others younger (85-)45(-25) Ma in Stevens et al. (2016: c.f. p. 1614 and Fig. 1A). Willyard et al. (2007) estimated upper (permineralized wood) and lower dates for divergence of the two subgenera of 85 and 45 Ma respectively (for the latter, see also Magallón & Sanderson 2005), although there were bouts of speciation much later. On the other hand, Millar (1998) suggested that this divergence and that of some of the sections had occurred by the middle of the Cretaceous. Crown-group Pinus is estimated to be (96-)89(-80) Ma by He et al. (2012; age rather similar in Gallien et al. 2015); the age of (80.1-)58.8(-45) Ma was suggested by B. Wang and Wang (2014: some estimates older), with most BEAST crown-group ages for both subgenera being 22-20 Ma, although some are as much as 50 Ma. Fossils of crown-group Pinus have been dated to 140-133 Ma (Ryberg et al. 2012; Falcon-Lang et al. 2016a, b, but c.f. Hilton 2016). Finally, W. T. Jin et al. (2021) suggested that stem Pinus was (177-)155.3(-135) Ma, and the crown age (116-)98.8(-83) Ma, so a phylogenetic fuse of ca 57 Ma here, but subsequent divergence did not occur for another 49 Ma or so towards the end of the Palaeocene.
Nuclear genome size in Pinus directly or indirectly correlates with a number of other features. Smaller genomes are associated with small seeds, wind (not animal) dispersal, invasiveness, fast growth, etc.; larger genomes are commoner in subgenus Strobus, which has genomes averaging ca 32.8 pg compared to those of subgenus Pinus at 27 pg (Grotkopp et al. 2004). For genome size - which can be very large - and evolution in Pinus, see Grotkopp et al. (2004). As in gymnosperms in general, rates of genome change are low, as are speciation rates, the two being correlated; Pinaceae have overall the lowest speciation rates among land plants (Puttick et al. 2015) in clades of any size, and diversification here way have something to do with temperature (J. B. Thompson et al. 2023).
Pinus now has a centre of diversity in Mexico and Central America, almost 50 species being native there, and of these ca 3/4 are endemic or practically so; Cupressaceae are also quite speciose there (Farjon & Styles 1997; Perry et al. 1998; Gernandt & Pérez-de la Rosa 2014). Klaus et al. (2017) suggested that ca 4.4 Ma is the estimated median node age in the genus.
Lockwood et al. (2013) dated diversification of Picea to the beginning of the middle Oligocene, only (37-)28(-21) Ma and Shao et al. (2019: transcriptomic analysis, dates for all nodes) suggested an age of ca 26.2 Ma, however, the oldest fossils in this clade are ca 136 Ma (Klymiuk & Stockey 2012) - a ca 100 Ma phylogenetic fuse (see also Leslie et al. 2018)? The genus probably originated in America and moved ca four times to the Old World via the Bering Land Bridge and thence twice to Europe (Shao et al. 2019); the rare P. breweriana, from the western U.S.A., is sister to the rest of the genus (Sigurgeirsson & Szmidt 1993: UPGMA analyses only; Shao et al. 2019).
The age of crown-group Abies has been estimated at (73.4-)48.6(-33.7) Ma (Xiang et al. 2014) or (24.5-)17.7, 16.2(-13.5) Ma (Semerikova et al. 2018) and ca 17.2 Ma (Z.-R. Wei et al. 2025), the last estimate with a phylogenetic fuse of around 50 Ma. In Abies there is weak support for the Californian endemic A. bracteata being sister to the rest of the genus while section Balsamea may be of hybrid origin (Xiang et al. 2014). Abies bracteata tended to be associated with a New World clade while the rest of the genus was a largely Old World clade; movement from the New to the Old World may well be responsible for the topological conflicts between the mitochondrial and the chloroplast plus nuclear trees (Semerikova et al. 2018).
Ran et al. (2018b) outline the distributions of a number of features within the two subfamilies; these have been incorporated into the characterizations.
Gene expression studies suggest that there is little in common between both the scaly structures of those Cupressaceae cones studied and the arils of Taxus with the ovuliferous scales of Pinaceae... (Englund et al. 2011; Groth et al. 2011). For a useful discussion of cone morphology and evolution in conifers s.l. and the complexity of the issues involved, see Matsunaga (2025).
Ecology & Physiology. For general information, see papers in Richardson (1998: Pinus), Andersson (2005), and especially Brodribb et al. (2012) and Augusto et al. (2014). Aspects of the ecology of Pinaceae along with that of other conifers is discussed briefly in Diversity & Distribution above and again more generally elsewhere, while details of ectomycorrhizal (ECM) associations are also discussed elsewhere.
Pinaceae are a notable ECM clade. The long-term environmental impact of these ECM relationships is of considerable interest, given the dominance of Pinaceae in the Boreal Forest biome and the important role they play in carbon sequestration there and elsewhere where they grow. Thus Augusto et al. (2014) date confirmed ECM symbioses in gymnosperms - these would be largely in Pinaceae - to the mid-Cretaceous some 115 Ma, probable ECM symbioses are dated to over 200 Ma in the Late Triassic, while possible ECM symbioses may have occured as early as in the Permian, over 250 Ma. At the same time Augusto et al. (2014) warn about extrapolating from the ecophysiological proclivities of modern gymnosperms to those of early gymnosperms.
Pinaceae currently dominate vast areas of mostly cool-temperate and Boreal forests in the northern hemisphere and they are the major component of the Boreal Forest/Taiga biome in particular (for which, see below. Estimates are that with aound a mere 615 or so extant species, conifers nevertheless make up over 39% of forests globally (W. T. Jin et al. 2021), so although they may not include many species, they are hardly unsuccessful. Single species dominate large areas in Boreal forests, although the number of species of conifers in general increases towards the tropics (see also Farjon 2017). Interestingly, this Boreal Forest/Taiga biome is very young, developing within the last 12 Ma or so as warm temperate broadleaf evergreen and mixed forests contracted with the drying and cooling of the climate then (Taggart & Cross 2009; Pound et al. 2012). Given the stressful abiotic environment of the Boreal Forest biome, it is trees with tracheidal tissue, relatively efficient as to wood construction costs yet "safe" in terms of water conductance (e.g. resistant to cavitation), that will be favoured (Hacke et al. 2005, 2015; Swenson & Enquist 2007). Fungus-plant relationships in such forests seem to be quite dynamic, thus looking at the relationship between ECM fungal diversity and increasing temperatures, forests along the boreal-temperate forest ecotones in North America will see a reduction in diversity while there is an increase in eastern temperate forests (Steidinger et al. 2018). In suitable conditions Pinus-dominated forests in particular occur much further south, the plants tolerating nutrient-poor soils and also quite dry climates, hence their current diversity in Mexico and their dominance in parts of the Mediterranean, and they also grow in Costa Rica (Janzen 1983) and even south of the equator, as in montane Sumatra (maps in White et al. 2000; Andersson 2005; see also Richardson & Rundel 1998 for Pinus). For the most part Pinaceae are unable to compete in tropical broad-leaved rain forests (but see the relatively broad-leaved P. krempfii: Brodribb & Feild 2008), while some species of Pinus in particular may not handle extreme drought conditions very well (Breshears et al. 2005; Stovall et al. 2019; see also elsewhere).
This association with ECM fungi is central to understanding the ecology of Pinaceae. As Read (1998: p. 328) noted, "pine roots are simply food bases which nourish an extremely dense mycelial system", the mycelium:root ratio being something like 200,000:1 on a soil volume basis. The ECM fungi aid in the uptake of both phosphorus and nitrogen, as well as helping the plants deal with the solubilization of toxic metals in the low pH soils that develop in pine-dominated communities (Read 1998; see also Scholes & Nowicki 1998). Similarly, Bahram et al. (2020) noted that in the Baltic area conifers drove a decrease in soil pH, an increase in the C:N ratio, a reduction in bacterial number and the bacteria:fungus ratio, an increase in the lignolytic plant cell wall degrading enzymes/carbohydrate-active enzymes ratio, and so on. For the complex interactions between fungal composition, species diversity, productivity, soil moisture and carbon content in Pinus sylvestris forests, see Hiiesalu et al. (2017). Pinaceae thrive in high-light conditions; they have high leaf mass per unit area and also a very high leaf area index, and although their seedlings have lower photosynthetic rates than those of angiosperms they tolerate leaf water stress better (Fu et al. 2012; see also Rundel & Yoder 1998). Longevity of pine and spruce needles increases in colder, northern latitudes (Picea needles may live for 12 years or more - Reich et al. 2014a). W. Chen et al. (2016) noted that the three ECM Pinaceae included in their study of nutrient uptake from the soil had thick to very thick first-order roots compared with those of other ECM plants and showed little response in terms of either ECM or rootlet proliferation in an environment where nitrogen was patchily distributed. They suggested that this may be because in places where Pinaceae normally grow the soil is more homogeneous and has large amounts of persistent leaf litter. N derived directly from rock can greatly increase ecosystem C storage in coniferous forests, and such forests grow in parts of the globe where the relative increase in total N coming from rock breakdown is highest, sometimes over 100% (Morford et al. 2011; Houlton et al. 2018). Conifers take up N both as NO3- and NH4+ (X. Zhou et al. 2020).
Estimates of live above-ground biomass in Pinaceae-dominated forests are in the order of 0.8-0.9 x 106 kg ha-1, that of Pseudotsuga menziesii perhaps even reaching 1.6 x 106 kg ha-1 (see Franklin & Dryness 1973). Figures for the total above + below ground C in the Boreal Forest/Taiga biome are about 505 Pg C, ca 34.2 kg C m-2, and a mean turnover time of (45.4-)53.3(-73.4) years (Carvalhais et al. 2014: tables S1 and S2). The fraction of biomass in the foliage decreases with latitude, that in the root tends to increase, both changes connected with the increased longevity of the needles and associated low values of new leaves produced annually in far northern conifers (Reich et al. 2014a: p. 13705 has it backwards; see Reich 2014b: Fig. 2). Accumulation of relatively non-flammable litter under genera like Tsuga and Picea can be massive, and it can also protect the trees against ground fires, while litter of Pinus, much more flammable, accumulates less (Cornwell et al. 2015 and references; for fires, see below). Wood of Pinaceae, as in other conifers, is broken down mostly by brown rot fungi. These fungi cannot degrade lignin, but readily break down cellulose and hemicellulose, leaving behind brown, crumbly detritus that is very rich in lignin and resistant to decay (Boddy & Watkinson 1995; Ayuso-Fernández et al. 2019; see above). Finally, it may be noted that mosses like Sphagnum are often common in the ground layer of the Boreal Forest/Taiga biome, and Sphagnum is a major accumulator of C in its own right.
Many Pinaceae tolerate burning, indeed, fossils of Pinus mundayifrom some 140-133 Ma, consist of charcoalified remains (Falcon-Lang et al. 2016a). A recent categorisation of burning intensities goes non-flammable—fast-flammable—hot-flammable. Fast-flammable includes fires of relatively low intensity, while the last class is associated with crown fires, the death of the plant, and serotiny (cones retaining mature seeds for more than a year - here the stimulus for seed releease is fire) or at least fire-stimulated germination (Pausas et al. 2017; Stevens et al. 2020); these are the resister and evader strategies in Jin et al. (2021). He at al. (2012) looked at the origin of fire-associated traits in Pinus, and found that thick bark characterised the whole genus, with its origin being somewhere between 126-89 Ma (absolute spreads greater), while very thick bark, branch shedding and serotiny were common in subgenus Pinus (hard pines), whose diversification was dated at (96-)89(-80) Mya (much older than other estimates - see also above, Jin et al. 2021; etc.); grass-like seedlings (see the next paragraph) were uncommon (He at al. 2012 noted that thick bark and serotiny were found in a few other Pinales). See also Turck et al. (2025) for adaptations to fires in conifers in general; some species of Pinus are resprouters (see also B. Wang et al. 2024), the fourth major adaptation to fire in conifers mentioned by Turck et al. In the mid Cretaceous the presence of shrubby angiosperms and ferns may have increased the prevalence of intense and rapidly-spreading fires that seriously affected conifer forests - indeed, traits for various aspects of fire resistance in Pinus seem to have evolved then - even if other conifer groups may have been driven to extinction (Gernandt et al. 2008; Belcher & Hudspith 2016).
Hot-flammable fires open the forest canopy, so making conditions suitable for species of Pinus subgenus Pinus in particular, many of which are adapted to fire-prone environments (Brodribb et al. 2012; Lamont et al. 2018a, b: see also Agee 1998; Schwilk & Ackerley 2001; Keeley 2012; Pausas 2015). Tierney et al. (2019) noted that the amount of nitrogen volatilized when longleaf pine savannas (replanted, restoration project) were burned was greater than that produced by N fixation, etc., so conditions under burning were becoming more oligotrophic, perhaps the original conditions for such forests (see also above). He et al. (2012; also Bond & Midgley 2012; Lamont et al. 2018a, b) thought that thick bark resistant to low intensity fires, the shedding of dead lower branches and so the development of long, naked trunks, would tend to prevent crown fires - this is the fast-flammable strategy of Pausas et al. (2017). These features evolved in Pinus around (147-)126(-105) Ma, and since this is the age of stem Pinus, it assumes that their evolution occured as the genus split from the [Picea + Cathaya] clade. Very thick bark, non-shedding branches and serotinous cones are an ecological syndrome adapted to high intensity (crown) fires (the hot-flammable strategy - Pausas et al. 2017), and this syndrome is dated to (96-)89(-80) Ma, the age of crown-group Pinus (He at al. 2012). Note, however, that whether or not there is serotiny may vary within a species, as in Pinus contorta (e.g. Benkman 1999). In some species of subgenus Pinus the juveniles have long and very dense needles covering the growing point, the so-called grass stage, and such seedlings tolerate fire; after the grass stage the stem elongates and thickens rapidly (Turck et al. 2025), while in species like P. palustris the adult sheds its branches. Fires facilitated in both cases are the fast-flammable type, both these seedlings and the adults surviving. Overall, character evolution here tends to be correlated (Schwilk & Ackerley 2001, etc.) - see also Proteaceae. Interestingly, although Pinus with its long needles has relatively well-aerated and very flammable litter like that of many/most other gymnosperms, leaves/needles of many other Pinaceae are smaller and the litter is compact and much less flammable (Cornwell et al. 2015), however, once it starts burning...; Stevens et al. (2020, esp. Fig. 2) discusses the variety of litter/flammability types in Pinaceae in particular and conifers in general. When the phylogeny of Pinaceae settles down, one can start thinking about the evolution of this set of traits - small needles and non-flammable litter are probably derived, but what about the situation in Pinus? See also Proteaceae in particular for adaptations to fires.
Associated with the tolerance of many Pinaceae to burning is the fact that a number of species of Pinus in particular are pioneer plants that regenerate soon after a burn, and the genus includes a number of invasive species that are aggressive colonizers after disturbance (Richardson & Rejmanek 2004). ECM suilloid fungi are notably common on Pinaceae, perhaps because the fungi establish ECM asssociations in early successional situations (Bruns et al. 2002; Hayward et al. 2015); ECM suilloid basidiomycetes include genera like Rhizopogon and Gomphidium. Suillus itself in particular is noted for its early association with Pinus, and its spores seem to be very widely dispersed (Hayward et al. 2015), and this association may be an element in the success of several species of Pinus as invasives (Policelli et al. 2019). However, the most invasive species of Pinus are often associated with most invasive ECM fungi like species of Amanita, Boletus, Inocybe and Paxillus, fungi that are involved in later-stage associations with Pinus (Policelli et al. 2022). Other factors are also involved, and the story is rather complex; I am unclear how any such transitions might occur.
Overall, ECM forests, and conifer-dominated forests in particular, show a fair amount of dynamism. Thus there are suggestions that recently Fagaceae may be replacing Pinaceae in some temperate forests (Alfaro Reyna et al. 2018). Here climate stress and successional dynamics may be involved, but changes are less obvious in eastern North America and in cases where there have been natural disturbances, and in any event there have not been significant changes in dominance... (Alfaro Reyna et al. 2018). However, oak does seem to be replacinmg pine in parts of Mexico (Alfaro Reyna et al. 2019). This issue is complex. Changes may be happening in part because Pinus in particular, along with ECM Populus, are early successional plants at higher latitudes, and they are replaced by other genera of Pinaceae; however, with increasing temperatures and associated fires this replacement may be retarded (Searle & Chen 2017). Along similar lines, Mack et al. (2021) suggested that fire may lead to extensive replacement of slow-growing black spruce, Picea mariana, by fast-growing species such as Populus tremuloides and birch, e.g. Betula neoalaskana, in western boreal forests in North America. I.a. this will affect C and N cycling in the community - for instance, much Picea-derived C will be converted to CO2 in the fires, but then C will be sequestered by the faster-growing angiosperm trees. Baltzer et al. (2021) flesh out this story, noting that in other parts of its range black spruce may be replaced by jack pine, Pinus banksiana; moisture deficits increase with global warming and fires increase, and in some situations black spruce shows little resilience. So sometimes Pinus becomes more common, and at the expense of other conifers.
Given the close association of ECM fungi and Pinaceae, knowledge of the age of the clades of both is of interest. Crown group ages for the origins of ECM clades of Agaricales were split about equally between the Late Cretaceous and Eocene, and for nearly all a Jurassic origin could be rejected; Pinaceae-associated fungi were linked with both the Eocene and the Late Cretaceous dates (Ryberg & Matheny 2012). The ECM\sebacina clade (= Sebacinaceae - Weiß et al. 2016) in Sebacinales seems to have evolved on North American temperate Pinaceae (87-)57, 45(-30) Ma (Tedersoo et al. 2014a: stem and crown fungal ages). Diversification rates in the basidiomycete Russula are highest in extratropical lineages/those associated with Pinaceae (Looney et al. 2015). However, many estimates suggest that Pinaceae had begun diverging long before these fungal ages (see above). Given current uncertainties over details of crown Pinaceae diversification, the relation between the evolution of ECM fungi and Pinaceae remains unclear, although the ECM association can be thought of as an apomorphy for the family.
There is discussion of the relationship between bark beetles and various species of Pinaceae in particular below. Bark beetles cause sometimes massive mortality in conifer populations in many parts of the world (Fettig et al. 2021: Fig. 1 for the western U.S.A.), and this affects fire incidence (Fettig et al. 2021 and references). Recently-killed conifers may be notably flammable as the composition of the needles is different from that of living needles and the needles dry out; beetles may also kill survivors of fires. Until recently, at least, conifer mortality caused by bark beetles has been greater than that caused by fires, although it would be interesting to see what the most recent figures look like; however, as mentioned above, a number of conifers can be considered to be adapted to fires. But even if the recently-killed trees are not burnt and so converted to CO2, the scale of the beetle infestations has a perceptible effect on the overall C balance. Over the years CO2 is released more gradually as the wood decays, but it is also taken up and temporarily sequestered by the replacement vegetation (Ghimire et al. 2015b).
As Bag et al. (2025: p. 420) note "Today's conifers [discussion centres on spruce and pine] are the dominating species only in ecosystems that impose significant plant stress – such as in the boreal forests, higher mountains, and dry and/or nutrient-poor conditions". There are problems for evergreen plants growing in boreal conditions in spring in particular when insolation and hence incident energy increase yet temperatures may be below freezing - energy input increases, yet carbon assimilation is low. Bag et al. (2025) describe how chloroplast thylakoids become unstacked, mechanisms for dissipating excess energy are activated, and so on.
Wilhelm (1883), see also Wulff (1898), noted that stomata in species throughout Pinaceae were blocked by wax deposits, presumably affecting transpiration. Jeffree et al. (1971; see also Wulff 1898), looking at Picea sitchensis, found that the wax tubules that filled the "stomatal antechambers" (they covered the epidermis of the stomatal band as a whole) reduced transpiration by about two thirds, but photosynthesis was reduced less, by one third.
Large stems of some Pinaceae are quite frequently found very close together, whether because of vegetative growth or because of the germination and establishment of a number of seeds from an overlooked animal cache (Tomback & Linhart 1990 and references).
Most species of the dwarf mistletoe Arceuthobium (Santalaceae-Visceae) parasitize various Pinaceae, with a few species also growing on Juniperus (Cupressaceae - Farjon 2008); they can be serious pests of Pinus in particular, e.g. A. vaginatum on P. ponderosa (Unger 1992; Harrington & Wingfield 1998).
Resins from Pinaceae are not often major sources of amber, but along with Sciadopitys, Araucariaceae and perhaps Cupressaceae they seem to be components of the Baltic amber (Seyfullah et al. 2018).
Living up to 4,844 years or so, the bristlecone pine, Pinus longaeva, is the longest-living non-clonal seed plant (Munné-Bosch 2014 and references; Piovesan & Bondi 2021); its needles, which can live for over 30 years (Hacke et al. 2015), are the longest-lived leaves of all land plants except Welwitschia - and the leaves of the latter are ever-growing. Pasques and Munné-Bosch (2022) looked at ageing in P. uncinata, from - here "old" individuals are only some 700 years old. They suggested that factors like loss of apical dominance, development of epicormic buds and modular senescence helped ensure the longevity of the plant. Polyphenolic parenchyma cells in the phloem of Norway spruce (Picea abies) can remain alive for over 70 years (Krokene et al. 2008). For more on age and plants, see also elsewhere, and consult Wikipedia, "List of Oldest Trees".
Pollination Biology & Seed Dispersal. See also above. Details of pollination mechanisms are quite diverse, although the pollen usually lands on the pollination droplet exuding from the micropyle and overall wind pollination is the norm (Little et al. 2014). Nepi et al. (2016, esp. 2017) found that the droplets coming from the micropyle of such wind-pollinated taxa were lower in sugar but higher in total amino acids than those of ambophilous (with both wind- and insect-pollination) taxa. J. Doyle and O'Leary (1935b) described the distinctive pollination in Larix and Pseudotsuga where the pollen, which lacks sacci, lands on an almost stigmatic extension of the integument, the margins of which tend to inroll; contact with the nucellus may (Larix) or may not (Pseudotsuga) be needed for pollen tubes to develop. C. G. Williams and Greenwood (2022) suggest that pollen in Pinus taeda, at least, is dispersed in raindrops, potentially over very long distances. Comparing the young seed cones of Picea and Abies, Losada and Leslie (2018) found differences in appearance of the cones could be understood in terms of variation in the rate of cone scale development, but it seemed to have little to do with how effective pollination was.
The female cones of several genera of Pinaceae are thermogenic, being up to 150 C warmer than the needles, and as a consequence they emit infrared radiation (Takács et al. 2017) - and for the consequences of that, see elsewhere.
For details of seed morphology, dispersal types, etc., and their evolution, see Contreras et al. (2016), Leslie et al. (2017) and Vander Wall (2023). Tissues of pine cones are mostly dead, but the ovuliferous scales open and close (the scales bend) because of a layered structure with a different orientation of cellulose microfibrils in the layers - there is an outer layer of sclereids and an inner layer of fibres, which are in bundles (Dawson et al. 1992: Pinus radiata; see also Poppinga et al. 2016). The shape of the wings in seeds of Pinaceae is largely determined by the shape of the cone scales (Diedrich & Leslie 2016). Cone scales in Pinaceae may reflex to allow the seeds to disperse, or the scales may fall from the cone, seeds falling out at the same time; the latter condition is commoner when the seeds are larger and are packed more densely in the cone (Losada et al. 2019). Seeds of species in subgenus Pinus tend to be smaller than those in subgenus Strobus; large seeds in the latter subgenus are associated with more arid environments - and this links on to vertebrate dispersal (Salazar-Tortosa et al. 2019); around 25% of the species in the genus lack wings. About 24 species of pine are serotinous, particularly those susceptible to crown fires (e.g. Hernández-Serrano et al. 2013 and literature, see also above). Pinaceae tend to show (weak) masting behaviour (Koenig & Knops 2000; Vander Wall 2023), and masting behaviour and serotiny may interact. Thus LaMontagne et al. (2021) found that variability in the time of masting was greater in species that took two years from bud primordium to seed maturity and less in serotinous species and also species in which the seeds took three years to mature (the latter - Pinus). Relative temperatures affected reproduction: Hot years followd by cooler year at the time of cone initiation led to higher reproduction in the case of Abies, Picea and Tsuga, while the reverse was true of Pinus (LaMontagne et al. 2021). See Fagaceae for more on masting. Note that, as suggested above, there is extensive variation in life cycles in Pinaceae. The time from pollination to fertilization may be over a year and pollen germination can take months (Little et al. 2014 and references), and the time from pollination to production of mature seed may be over two years (Owens et al. 2003) - and this is independent of serotiny.
We turn now to seed predation and dispersal. Note that some species of Pinus are animal dispersed, others are both animal- and wind-dispersed, i.e. they are diplochorous. Seeds of ca 20 species of Pinus (nearly all subgenus Strobus) lack wings and are primarily dispersed by nutcrackers and other corvids, and such seeds tend to be larger than those of their winged relatives - from which they have evolved four to twelve times (Contreras et al. 2016; see also Tomback & Linhart 1990; Leslie et al. 2017). Spotted nutcrackers Nucifraga caryocatactes, are the sole seed dispersers of the Swiss stone pine, P. cembra (subgenus Strobus), and juveniles (1 year old) of this species show more variation in how they disperse the seeds of the pine than do adults (2 years old or more), including having an overall greater dispersal range and caching seeds at higher altitudes (Graf et al. 2025). Seeds of lodgepole pines (Pinus contorta, subgenus Pinus) in North America are commonly dispersed by red squirrels (Tamiasciurus), which eat the seeds or cache the cones (see especially C. C. Smith 1970), and crossbills (Loxia curvirostra complex, the rank of the entities involved is not always clear), which pry the seeds of cones remaining on the tree loose (Thompson 2005). Note that there is infraspecific variation in P. contorta, and in the more humid conditions west of the Cascades the species is not serotinous and there may be crop failures, while east of the Cascades serotiny is common and cone production more reliable; there are different species of squirrel in the two areas. Normally, pine cone morphology is adapted to the presence of squirrels, the cones being long and rather narrow, and the scales being distally thinner, but in some isolated areas where the pine grows but there are no squirrels there has been local selection on cone morphology by the crossbills, and the bill shape of the crossbill varies along with cone morphology (Benkman 1999; Thompson 2005; Benkman 2025). Interestingly, there is comparable variation involving red squirrels, crossbills and black spruce, Picea mariana, in eastern North America (Thompson 2005); crossbills also eat the seeds of Tsuga, Pseudotsuga and other genera although, while corvids (nutcrackers, Nucifraga), pines and squirrels similarly interact (Benkman 1999). Some figures for seed dispersal are staggering. One nutcracker may store some 3,200 seeds/hectare - and this is just for one species of pine in one season (Tomback & Linhart 1990). Clark's nutcracker, Nucifraga columbiana, can carry up to 93 pine seeds in its sublingual pouch, and fly up to 32 km or so before caching seeds (Vander Wall 2023). For the relationship between pinyon pines, Pinus edulis, and Clark's nutcracker, see Vander Wall and Balda (1977), and for that between the pine and the pinyon jay, a flock of 250 of which can cache around 4.5 million pine seeds per year, close to 20,000 per bird(!), see Ligon (1978). Corvids in particular have remarkable memories for where they cache seeds, but their sense of smell is poor, but a number of vertebrates have a very good sense of smell and find cached seeds particularly after rain (Vander Wall 2023). And what about the age of such interactions? Crossbills probably split from their relatives, redpolls, a mere 750,000-1.4 Ma, or perhaps as much as some 6 Ma (Arnaiz-Villenor et al. 2001; Benkman 2025), but in either case far younger than ages commonly associated with the diversification of Pinus (see above).
Plant-Animal Interactions. Iason et al. (2011) tested monoterpenes, common in pines, for protection against herbivory by capercaillie, bank voles, slugs, or red deer; some, but not all, worked (see also Hamberger et al. 2011: defensive properties of diterpene resin acids). Pine needle phenolics and pine stem resins are constitutively more abundant in Nearctic than in Palaearctic Pinus, but there was no difference between the two groups in how inducible these defences were (Carrillo-Gavilán et al. 2014: seedlings examined). Mumm and Hilker (2006) discuss the chemical defence of pines against foliovores in particular; for conifer exudates, see Lambert et al. (2007a). For the antifeedant effects of piperidine alkaloids, see Virjamo and Julkunen-Tiitto (2017). The gut microbiota of the pine weevil Hylobius abietis degrades protective diterpene acids of Picea abies, and interestingly, it is similar to that of other species of Hylobius, also sawflies, etc., all with a similar life style (Berasategui et al. 2017 and references). For a summary of insect-pine interactions, see de Groot and Turgeon (1998) and in particular the papers in Vega and Hofstetter (2015).
Some bark beetles in general (Curculionidae-Scolytinae and -Platypodinae), and ambrosia beetles in particular (see next paragraph) (Wood 1982; Wood & Bright 1992; Six 2012; Vega & Hofstetter 2015; Huler & Stelinski 2016), highly derived weevils, have a very close association with Pinaceae (Jordal et al. 2011; see also Gohli et al. 2017: Fig. 1). Bark beetles include some 3,700 species that make their complex gallery systems often in the phloem, and members of genera like the North American Dendroctonus and the Northern Hemisphere Ips can be very noxious pests, a few invading living pines (e.g. Franceschi et al. 2005; Six 2012; Ramírez-Reyes et al. 2023: features of Dendroctonus and its infestations mapped on to beetle phylogeny). Dendroctonus may have evolved ca 52.4 Ma, a clade all members of which live on Pinus in tun diversifying ca 48.6 Ma, most speciation there being in the Miocene when pines were also diversifying (Ramírez-Reyes et al. 2023). For the southern pine beetle, D. frontalis, see Sullivan 2011; Lesk et al. 2017: future spread). Although the beetles tend to attack relatively few species of conifers (they are mono- or oligophagous), outbreaks can be devastating, beetles being attracted to trees by pheromones produced by beetles that are already there, pheromones that - ironically - may be slight modifications of, for example, terpenes produced by the tree being attacked (e.g. Raffa et al. 2015). In such situations the exploding beetle population may be able to overwhelm the defences of even healthy trees (Kelley & Farrell 1998 for host specificity; Franceschi et al. 2005; Sullivan 2011; Raffa 2014; Raffa et al. 2015; Weed et al. 2015; Celedon & Bohlmann 2019; Koontz et al. 2021). Drought conditions may also make the trees more susceptible to attack (see Huang et al. 2019 and references), thus drought and tree size interacted to produce devastating infestations of D. brevicomis on Pinus ponderosa in the Sierra Nevada, California (Koontz et al. 2021), however, tree size was not always important in such attacks, and other factors are also involved (Netherer et al. 2015), for instance, unhealthy trees have fewer rewards for the beetles than do healthy trees. Bristlecone pine, P. longaeva, has higher levels of constitutive expression of potential defensive compounds like (+)-α-pinene in its phloem unlike the other pines growing with it - it has over twenty times the amount of that in limber pine, P. flexilis, for example - and it is not attacked by Dendroctonus (Bentz et al. 2016; see Piovesan & Bondi 2021). Hammer et al. (2020) found that herbivory by D. frontalis on P. caribaea was reduced when the concentration of the phenylpropanoid 4-allylanisole was over 2.5% in the xylem resin, but this can be reduced if diluted by monoterpenes. However, as Raffa (2014) notes, the relationships between the mountain pine beetle D. ponderosae and pine, for instance, are extremely complex and context-dependent, and how the beetle will react to pines such as the white pine, P. albicaulis, that it encounters as its range changes is not easy to predict. Certainly, introduced bark beetles tend not to cause serious damage in their new homes, in part perhaps because of their host specificity (Forgione et al. 2022). Many aspects of these relationships are mediated by terpenoids, whether produced by the weevil as pheromones or by the pine (predominantly mono- and diterpenes, but also some sequiterpenes), importantly, terpenoid concentrations are affected by a variety of bacteria which metabolize particular terpenoids, apparently using them as a carbon source. Furthermore, enemies of the beetles are attracted both by plant volatiles induced by herbivores and by beetle pheromones (Raffa 2014). The pine weevil Hylobius is an important pest of conifers in Europe, the adult in particular killing seedlings and saplings, and its gut microbiota thrive on the diterpenes the weevil ingests, increasing the number of eggs it can lay (Berasategui et al. 2017). Nitrogen-fixing bacteria are associated with the bark beetle Dendroctonus (Berasategui et al. 2017 and references). Bark beetles in general eat a variety of tissues, although most scolytines eat phloem (see Kirkendall et al. 2015: T. 3.1 for a classification of feeding behaviours), and as mentioned their microbial symbionts can metabolise pine terpenes, so neutralizing one of the trees' defences.
Ambrosia beetles are scolytine and platypodine weevils (note that details of the relationships of Platypodinae are unclear - Hulcr et al. 2015) that are fungus farmers ("xylomycetophagy"). They are associated with blue-stain fungi, mostly ascomycetes such as Ophiostoma and Ceratocystis (Ophiostomatales and Micrascales respectively, both sordariomycetes), some yeasts (e.g. Rivera et al. 2009), as well as a few basidiomycetes, although there is a diversity of other associates including bacteria, parasitoids of the beetles, and fungus-eating nematodes, and also mites which may carry spores of the fungi from tree to tree (Hofstetter et al. 2015). Some of the fungi associated with the beetles may be pathogenic, and include Ophiostoma, which i.a. causes Dutch elm disease. There have been at least five origins of fungal farming in ascomycetes, two or more in basidiomycetes (Hulcr & Stelinski 2016, see also Diehl et al. 2022 for farming). These fungi effectively circumvent some bark defences (see below) since they hitch rides in the mycangia of the beetles, intricate cuticular invaginations in which they carry a fungus inoculum that will infect the tree, and the beetles tunnel through the bark (Krokene et al. 2008 and references). The fungi grow in the sapwood and sometimes heartwood and help hasten the death of the infected tree (Franceschi et al. 2005); the fungus is vertically transmitted and fully domesticated (Vanderpool et al. 2017). The beetles, some 3,400 species, mostly tunnel in dead or dying wood, although early-branching members of the ambrosia beetle clade may live in phloem; both beetle larvae and adults obtain all their nutrition from the fungi they farm. The larvae eat yeast-like bodies proliferating from the cultivated fungi in the beetle galleries (Jordal et al. 2000; Cognato et al. 2011 and references); the mouth-parts of the weevil are also much modified. Interestingly, females of the beetle Xylosandrus germanus preferentially attack trees that have high ethanol, a product of anaerobic respiration that is particularly abundant in stressed plant tissue. Its fungal associates like Raffaelea and Ambrosiella (more sordariomycetes) can detoxify alcohol, and they also produce it. However, ethyl alcohol is toxic to the possible competitors of these fungi such as Aspergillus in the beetle galleries (Ranger et al. 2018). Trees suffering from stress, as well as recently-sawn timber, are the prime targets of the weevils (Hulcr & Stelinski 2016). Overall, angiosperm hosts are more common, and the evolution of the ambrosia feeding habit, which has happened 8 times or more in the weevils (ca 15 or more times - Hulcr & Stelinski 2016; see also Jordal & Cognato 2012), is associated with shifts to using angiosperms (Six 2012); development of fungus cultivation is unreversed (Beaver 1989; Farrell et al. 2001; Mueller et al. 2005; Jordal et al. 2008 and references, 2011; Jordal & Cognato 2012; Gohli et al. 2017). Most of the origins of farming have been in Scolytinae, one in Platypodinae, of the latter, a clade of ca 1,400 weevils, all but some basal clades are farmers (Jordal 2015). Hylurgini (scolytines, used to be Tomicini) weevils found on pines are in a separate clade from those found on Araucaria (Jordal & Cognato 2012); see also Wood (1982), Wood and Bright (1992) and Vega and Hofstetter (2015) for the weevils. Platypodinae are ca 119 Ma (stem age) or 88 Ma (crown age) (Jordal 2015), the scolytine associations are younger (Vanderpool et al. 2017). The oldest fossil bark beetle known is from Lebanese amber ca 129 ma (Kirejtshuk et al. 2009).
Despite the defences of pines, etc., the blue-stain fungi that are carried by the beetles can quickly invade the sapwood and render it non-functional, basically clogging up the phloem and so killing the plant, sometimes quickly, sometimes quite slowly. Some, at least, of these fungi (e.g. Endoconidiophora polonica on Picea abies, Norway spruce), detoxify the plant's defences against the weevil Ips typographus by beginning the breakdown of stilbenes (in the phenolic defences) and flavonoids (in the resin), i.e. in the two major components of the plant's defences (S.-H. Li et al. 2012; Keeling & Bohlmann 2006a). Genes involved in the synthesis of such defence compounds are expressed in the infected pine, but nevertheless the amounts of the products decline as they are used up by the fungus (Wadke et al. 2016), the defences against the weevil carrying the fungus being neutralized, the phenolic defences being used by the fungus as a carbon source (DiGuistini et al. 2011; Wadke et al. 2016). Along similar lines, R. Sun et al. (2025) found that I. typographus attacking P. abies can hydrolyze phenolic glucosides in the spruce, producing the corresponding aglucones, and these aglucones have increased antifungal activity; in this case the aglucones protect Ips against the entomopathogen Beauveria/Cordyceps bassiana (an ascomcycete, Hypocreales; it causes white muscardine disease). However, B. bassiana can detoxify stilbene and flavonoid aglucones in the weevil by forming methylglucoside derivatives that are not toxic to the fungus.
Understanding the details of the evolution of the defence system against such weevils in part depends on our knowledge of Pinaceae phylogeny (e.g. Hudgins et al. 2004), and this is still somewhat unclear. Hudgins et al. (2003, 2004) examined the diversity of bark beetles in conifers in the context of various plant structures that might be defences against such beasts. The beetles eat the wood despite the resin canals in the phloem of Pinaceae in general and also in the xylem of Pinoideae (e.g. Hudgins et al. 2004; Franceschi et al. 2005; Krokene 2015), although paradoxically Cupressales, which have only sporadic resin canals, nevertheless harbour a lower diversity of these beetles. That issue aside, the gut flora of the spruce weevil Pissoides strobi on resistant Picea sitchensis seems not to flourish (Whitehill et al. 2018), but the chemical defences of conifers can sometimes be circumvented by the activities of these bacteria (Berasategui et al. 2017). Furthermore, stone cells, intracellular crystals, phenolics in phloem-associated polyphenolic parenchyma (PP) cells, etc., may also be part of the defences. Stone cells in particular may be similar to the sand defences of some Nyctaginaceae where sand grains stick to glandular hairs; the stone cells/sand grains both wear down insect mandibles and perhaps make the plants generally distasteful (Whitehill et al. 2018, see also Lopresti & Karban 2017). Keeling and Bohlmann (2006a, esp. b; see also Celedon & Bohlmann 2019) describe terpenoid diversity (monoterpenes, sequiterpenes) and conifer defence mechanisms, a complex subject; it is unclear just what is responsible for the considerable diversity of terpenoids that are found here, although multisubstrate and multifunctional enzymes involved in terpenoid synthesis in Picea sitchensis (Sitka spruce), for example, may well be involved (Hamberger et al. 2011). In Abies grandis (grand fir) a single terpene synthase, γ-humulene synthase, is involved in the synthesis of 52 different sesquiterpenes (Steele et al. 1998), and similar sesquiterpenes were both constitutive and produced in response to wounding.
The tortricid moth Choristoneura has twice moved from angiosperms, where it is a generalist feeder, on to Pinales, where pines and spruce are its favourites and where it can cause serious damage. Indeed, endophytes of Picea (spruce) produce several metabolites toxic to the eastern spruce budworm C. fumiferana (J. D. Miller et al. 2002; Findlay et al. 2003; Sumarah et al. 2010). These shifts have been dated to ca 11 Ma, although divergence in the North American spruce budworm complex, one of the clades involved, has been dated to to a mere ca 3.5 Ma (Fagua et al. 2018). Pinus is an important host plant for lepidopteran larvae in the contiguous United States of America, being included in the top 10 such plants; Picea and Pseudotsuga are also quite important hosts (Narango et al. 2020). The lasiocampoid Dendrolimus is another serious foliovore; for a phylogeny, see Qin et al. (2019: "direct contact with living or dead caterpillars, even their pupae, results in poisoning known as caterpillar arthritis"). Leaves of the Douglas fir, Pseudotsuga menziesii, are apparently often eaten by the gelechiid moth, Chionodes.
Sawflies, i.a. Diprionidae, are sometimes serious pests of Pinaceae (Schneider 2016), and different species of these insects may eat leaves (the larvae are gregarious and can defoliate the plant) or bore in wood, and defences against them are discussed by Celedon and Bohlmann (2019).
Turgeon et al. (1994) noted that ca 85% of the insects recorded as eating conifer cones had been recorded from Pinaceae, but that was probably due to the economic importance of the family; they also noted that almost as many hymenopteran parasitoids of these insects had also been recorded. Of the seed predators, Megastigmus (Hymenoptera-Torymidae) is perhaps most important; it lays its eggs in the ovules before fertilization (c.f. Cupressaceae: Roualt et al. 2004). The female cones of several genera of Pinaceae, apparently thermogenic and up to 150 C warmer than the needles, emit infrared radiation, so making the cones very conspicuous and attracting a hemipteran bug, Leptoglossus occcidentalis, which has IR receptors - it is a seed predator (Takács et al. 2017).
Some 70 species of Adelgidae (aphids) are restricted to Pinaceae, and they include woolly adelgids such as Adelges/Dreyfusia/Chermes piceae and A. tsugae, serious introduced pests in North America, the former being a major pest of fir, Abies spp., including A. fraseri, and the latter of hemlock, Tsuga canadense (Timell et al. 1986: vol. 3, chapter 20; Havill et al. 2007; Kaylor et al. 2016: difficulty in predicting the fate of A. fraseri). In A./D./C. piceae there are five different generations in a single life cycle, the sexual, gall-forming generation being on Picea; as with other aphids, vertically transmitted bacteria are part of this ecosystem (Havill & Foottit 2007). Cinara, a genus of lachnine aphids with about 250 species, radiated on conifers beginning ca 50 Ma (Meseguer et al. 2015; ) or (88.5-)78.5(-68.5) Ma (R. Chen et al. 2016). It and other eulachnines (all told, ca 290 species) grow only on conifers, but they are embedded in a clade in which the original hosts may have been angiosperms (R. Chen et al. 2016) - note, however, that Peccoud et al. (2010) suggest that the orignal hosts of Aphididae as a whole may have been conifers. Cinara itself is found mainly on Pinus, perhaps its original host, and also Abies and Picea, although some species feed on Cupressaceae (Meseguer et al. 2015; R. Chen et al. 2016). Cinara species live mostly in the bark while other eulachnines feed on needles of Pinaceae alone (R. Chen et al. 2016). For speciation of Cinara, where closely-related species are found on different hosts, see Favret and Voegtlin (2004). In genera like the pemphigine Procophilus the secondary host of the aphid is on Pinaceae, where the insects move down to the roots, fossil aphids perhaps belonging here are known from Baltic Amber 48-34 Ma (Hille Ris Lambers 1979; Seyfullah et al. 2018).
Cecidomyiid gall midges are quite common on North American members of the family (Gagné 1989). See Powell et al. (1999) for other insect-conifer associations.
Many of the wood-eating basal termites (they have protozoans in their guts that can break down lignin) seem to like the wood of Pinales; the crown-group age of termites is Jurassic, ca (170-)149(-136) Ma (Bourguignon et al. 2014).
Plant-Bacterial/Fungal Associations. ECM associations are common in Pinaceae, and appropriate ECM fungi may have to be introduced if Pinus, for instance, is to be grown successfully in areas in which it normally does not grow (see Hayward et al. 2015: not always necessary; Vellinga et al. 2009: ECM introductions in general, over half on Pinaceae-Pinus). Both Suillus and Rhizopogon are notable ectomycorrhizal associates of Pinaceae, species of the former in particular tending to associate with different species/groups of species in Pinus or genera in Pinaceae, indeed, in some cases invasions of Pinus are achieved only by coinvasions with their particular Suillus associate (Lofgren et al. 2024). The beneficial effects of suilloid boletes such as Suillus and Rhizoderma are particularly evident (Hoeksema et al. 2019: meta-analysis; Sato & Toju 2019 for a phylogeny of boletes). Overall ECM fungal diversity may be low in early successional situations when suilloids establish associations with pines, but this is by no means always so for pine-ECM communities (D. L. Taylor et al. 2013; Anderson et al. 2013). Garcia et al. (2015 and references) suggest that details of the establishment of ECM associations may differ from those in flowering plants, genes of the common symbiotic signalling pathway (CSSP) (for both ECM and AM associations) not being involved. The relationship between Suillus and Pinales is discussed by Lofgren et al. (2020, 2024), the original host of Suillus probably being Larix, and particular species of the fungus usually form associations with particular genera of Pinaceae, or subgenera of Pinus, the associations being mediated by secondary metabolites produced by the fungus. Kernaghan and Patrinquin (2011) suggest that ECM suported by Larix are very specific, although there seems to have been little work carried out on these associations; Molina et al. (1992) think that there are a number of fairly close associations and Pinaceae, for instance, a large number of fungi associated with Pseudotsuga menziesii are apparently specific to it. There is further discussion on fungal specificity in Pinaceae in Molina et al. (1992).
As in other ECM associations, the complexity of pine-ECM fungus networks may be considerable (Simard et al. 2012). Thus Simard et al. (1997) found that ca 6.6% of the carbon fixed by Betula papyrifera moved to Pseudotsuga menziesii via their common ECM associate, while 5-15% of fixed 15N2 moved from Alnus glutinosa to Pinus contorta (Ekblad & Huss-Danell 1995), although the ecological significance of the latter is unclear. Associations are not fixed. Thus in central Italy several extra-zonal basidiomycete fungi that are usually associated with Pinales, now no longer growing in the area, have become associated with Arbutus unedo (Di Rita et al. 2020). Fungi associated with Ericaceae-Arbutoideae in western North America may also be ECM associates of Pinus (Molina et al. 1992; Bruns et al. 2002; Kennedy et al. 2012 and references), and such connections have important implications for forest regeneration. ECM associates of Picea may simultaneously be ericoid mycorrhizal associates on Ericaceae, with carbon and nitrogen moving between the two plants (Grelet et al. 2009, 2010), and fungi that form ericoid mycorrhizae can be endophytic in Picea (Vohnik et al. 2013). The oldest and the youngest individuals in Pseudotsuga menziesii forests may be linked by ECM networks (Belier et al. 2010), although this may preferentially benefit the larger individuals (see Weremijewicz et al. 2016: there are similar phenomenon in AM associations).
Pinus and Larix in particular may also form ectendomycorrhizal associations with ascomycetes (Peterson 2013). Recent work shows that a single species of ascomycete can form ERM (ericoid mycorrhizal), endophytic and ECM associations, the latter two in particular with Pinaceae (e.g. Vrålstad et al. 2000, 2002; Grelet et al. 2009; Perotto et al. 2018; Martino et al. 2018), growing in the same habitat, indeed, Ericaceae and Pinaceae often grow together. Furthermore, dark septate endophytes, ECM and ERM commonly have melanin, a substance particularly resistant to degradation, in their hyphae (e.g. Butler & Day 1998; Read et al. 2004; Bardgett et al. 2014; Clemmensen et al. 2014; Peter et al. 2016; Lindahl & Clemmensen 2017; Martino et al. 2018), and so C in these hyphae turns over more slowly than that in AM hyphae (Phillips et al. 2013; Fernandez et al. 2013, 2016; Fernandez & Kennedy 2018). Fernandez and Koide (2014) found that amounts of melanin, along with those of N, determined the rate of hyphal breakdown, suggesting that from this point of view melanin was an analogue of lignin, being notably decay-resistant (see also Fernandez et al. 2016). Bjorbækmo et al. (2010) and Timling & Taylor (2012) noted the high frequency of melanized fungi and dark septate endophytes in high northern latitudes where Pinaceae often dominate, melanin being an important component of the sequestered carbon in at least some older boreal forests (Read et al. 2004; Clemmensen et al. 2014; Fernandez et al. 2014, 2016; Lindahl & Clemmensen 2017).
Interestingly, there is quite extensive evidence that the particular ECM (and other) associates of individuals of a particular species of conifer may depend on the genotype of the latter. Thus the genotypes of seedlings of piñon pines (Pinus edulis) varied, but resembled those of their parents, which were either drought-tolerant, the basidiomycete Rhizopogon roseolus being prominent in their ECM community, or drought-intolerant, two species of the ascomycete Geopora then being prominent (Gehring et al. 2017a). There may be extensive mortality of ECM conifers, including P. edulis, under drought conditions (Breshears et al. 2005; McDowell et al. 2016; Stovall et al. 2019), and under such conditions P. edulis, for instance, would be replaced by the arbuscular mycorrhizal (AM) Juniperus (Gehring et al. 2017b). This has additional consequences, since litter of the AM juniper has a lower C:N ratio than that of the ECM P. edulis and decomposes faster (Gehring et al. 2017b and references), and there are also differences in root characteristics, e.g. in fine root diameter - rather narrower in ECM plants here (Valverde-Barrantes et al. 2017). Redmond et al. (2015) discussed the importance of nurse trees for young plants of the pine. For additional literature, see e.g. Schweitzer et al. (2004), Sthultz et al. (2009a, b), Gehring et al. (2014) and Gallart et al. (2017: form in which N is present also important). See also Brundrett (2017a), Tedersoo (2017b) and Tedersoo and Brundrett (2017) for literature, etc.; Larter et al. (2017) note physiological similarities in the AM Juniperus/ECM Pinus edulis comparison and that between Eucalyptus, often ECM, and AM Callitris species in Australia. There is more discussion under Ecology & Physiology above.
Bacteria associated with tuberculate ECM on Pinus contorta, a particular kind of ECM in which a cluster of root tips is surrounded by hyphae, are thought to fix nitrogen (Paul et al. 2007). Strains of Bradyrhizobium are the dominating bacteria in pine forests across North America, and although they are unable to fix nitrogen, they seem to be able to metabolize aromatic carbon sources (VanInsberghe et al. 2015). Some Pinaceae have foliar bacterial endophytes that fix nitrogen (Carrell & Frank 2014). In a final wrinkle of the story of how Pinaceae may acquire nitrogen, Laccaria bicolor, a brown rot fungus, took up nitrogen from springtails that it had first immobilized, and this nitrogen could be transferred to seedlings of Pinus strobus (Klironomos & Hart 2001).
A number of rusts, including those on ferns, have their aecial stages on Pinales, especially Pinaceae (Savile 1979b; Durrieu 1980). These include the white pine blister rust, Cronartium ribicola (alternate host Ribes, Grossulariaceae), introduced to North America around 1900 and a very serious pathogen of white pine, Pinus albicaulis, and its relatives (subgenus Strobus), and the activities of the fungus, along with those of the mountain pine beetle, mean that white pine no longer grows in large areas that it used to, and it has become a critically-threatened species (Harrington & Wingfield 1998; Stevens et al. 2016; Neale et al. 2024).
In Pinus strobus endophytes synthesize antifungal metabolites, effective against Microbotryum violaceum, parasitic on some Caryophyllaceae (Sumarah et al. 2010, 2011), and endophyte metabolites in spruce may be toxic to insects (Findlay et al. 2003). However, on occasion dark septate endophytes can reduce the growth of their hosts, although the relationship between the two is complex, being affected by temperature, host identity, whether or not the host has established a relationship with ECM fungi, etc. (Reininger & Sieber 2012 and references).
Harrington and Wingfield (1998) discuss a number of fungal diseases that can seriously affect the growth of species of Pinus.
Genes & Genomes. Genomes can be very large, as in the 19.6 Gb genome of the Norway spruce (Picea abies, Nystedt et al. 2013), even if intron length is only ca 2/3 that in Sapria (Rafflesiaceae: Cai et al. 2021); genomes in Pinus can reach 27.6 Gb (Y.-E. Lin et al. 2025). The rate of change of genome size is faster in Pinaceae than in other gymnosperm groups (Burleigh et al. 2012). Transposable elements (LTRs - Long Terminal Repeats - retrotransposons, perhaps especially Gypsy elements) may make up around 75% or even more of the genome in Pinaceae (Neale et al. 2014, 2024: q.v. for species with sequenced gnomes; Stevens et al. 2016). This large and extensive TE population may be (mostly) old, but there are no mechanisms for its removal (c.f. Nystedt et al. 2013).
Roa and Guerra (2018) note that 45S rDNA sites in Pinaceae tend to be in the middle of the short arms of the chromosomes, unlike in other gymnosperms, where they tend to be at the end and at the centromere, or angiosperms, where they tend to be near the end. For the nuclear karyotype, see Murray (2013); extensive synteny in Pinaceae persists for a long time, around 120-140 Ma (Pavy et al. 2012; de Miguel et al. 2015). Pavy et al. (2012) found that gene duplications were mostly very ancient, predating the angiosperm-gymnosperm split, again emphasizing the stability of the genome - the stasis here would be more than 300 My. This stability is reflected in the morphology of Pinaceae, their slow rates of speciation, etc., and may also be connected with their long-lived nature and large population size, while the high synteny and genome colinearity would facilitate hybridization (Pavy et al. 2012). Miguel et al. (2015) thought that the ancestral genome in Pinaceae was x = 12, 20 ancestral chromosome blocks having been shuffled around - there might be extensive synteny now, but this stasis had been preceded by a period of chromosome rearrangement. Nkongolo and Mehes-Smith (2012) discussed karyotype evolution in Pinaceae - i.a. they suggested that the karyotype of Pinus is ancestral to that of the group as a whole. There is extensive duplication of the knox-1 gene within Pinaceae, at least, although more general sampling is needed to pin down the point at which this duplication occurred (Guillet-Claude et al. 2004). Romanova et al. (2021) note that there was YABBY transcription family expression all around the leaf of Pseudotsuga menziesii and in its vascular tissue; the significance of this is unclear given the poor sampling for this transcription factor family. General aspects of nuclear genome evolution in vascular plants are discussed elsewhere.
Braukmann et al. (2009) chart the extent of the loss of ndh genes (see also Cronn et al. 2008; Hirao et al. 2008), and these genes are also lost in Gnetales - note that some details of the loss of nuclear transcripts encoding NDH proteins differ between the two, and there is also variation in Pinaceae themselves (Ruhlman et al. 2015). However, what is going on with ndh genes here (and in Gnetales) is unclear. In angiosperms such genes are most likely to be lost in plants that live in equable environments (and so in aquatic plants, for example), and in parasites, carnivores, etc.. However, the gymnosperms just mentioned include extremely long-lived plants growing in very stressful (arid) environments (think also Welwitschia), and as Sabater (2021: p. [8]) noted, "needles and the whole plant in several Gymnosperms do not appear to show PCD [programmed cell death] but show aging by accumulation of hazardous damages"; interestingly, Nicotiana mutants lacking ndh genes also seem to lack PCD, and in Viscum album, also lacking such genes, the leaves fall from the plant when still green (Schröder et al. 2022).
Z. Li et al. (2015) and Li and Barker (2019) suggested that there had been a genome duplication in the Picea area or even shared by all Pinaceae (see also J. W. Clark & Donoghue 2018), but Nystedt et al. (2013) and Zwaenepoel and Van de Peer (2019) doubt its existence.
The chloroplast inverted repeat (IR) is very much reduced in size in Pinales (e.g. Strauss et al. 1988; Raubeson & Jansen 1992; Tsudzuki et al. 1992; Lackey & Raubeson 2008; Jansen & Ruhlmann 2012), and Labiak and Carol (2017) talk about its drastic reduction or complete loss, while C. S. Wu et al. (2011a: p. 310) noted that the cpDNAs of Pinaceae "have preserved a rather reduced pair of IRs (236-495 bp) containing only the 3'psbA and trnl-CAU genes", ndh genes and one copy of the ycf2 gene being lost early on (Wakasugi et al. 1994; Wu et al. 2011a); as in Cupressales, there are small IRs (Guo et al. 2014). Interestingly, there is only a single form of the plastome in Pinus and Picea (W. Wang & Lanfear 2019: c.f. Cupressales). Qu et al. (2017) Note that different copies of the IR have been lost in Pinaceae and Cupressales (C.-S. Wu et al. 2011b). Associated with this IR (near) loss, plastome rearrangements are notably extensive in Pinaceae (Lin et al. 2010; Wu et al. 2011a, b), and Wu et al. (2011a) discuss inversions (two) in the plastomes, c.f. Cupressaceae in particular (Wu & Chaw 2016).
For biparental plastid inheritance, see references in Sullivan et al. (2017). Heteroplasmy is reported (see C. Lee et al. 2020).
Sullivan et al. (2019) found that there were highly recombinogenic small repeats in the large and overall very recombinogenic mitochondrial genome of Picea abies, but these are found not uncommonly in other vascular plants. Mitochondrial transmission is maternal in Pinus (Neale & Sederoff 1989; X.-R. Wang 1996; Adams 2019 and references); B. Wang and Wang (2014) discuss the complex history of mitochondrial inheritance in the genus.
Economic Importance. The majority of the world's lumber comes from softwood, and most of that comes from members of Pinaceae, and this despite the small size of the family (Mullin et al. 2011 and references). Interestingly, over 20% of the species of Pinus alone are invasives, P. radiata, with a narrow native range, being a good example, although it is also planted (Lavery & Mead 1998; Gallien et al. 2015), and this is facilitated by their ability to form ECM associations with Suillus species whose spores are very widely dispersed (Hayward et al. 2015; Peay 2016; Lofgren et al. 2024).
Pinaceae, species of Pinus and Tsuga in particular in North America, can be very heavily infested by bark beetles, a variety of fungi, tortricid moths, dwarf mistletoes, etc. (see above: Plant-Animal Interactions), that kill the plants, especially when they are stressed (e.g. de Groot & Turgeon 1998; Harrington & Wingfield 1998). The effects of these organisms are exacerbated by the tendency of conifers to be locally very abundant, and some herbivore- and pathogen-induced die-offs have been very extensive. Several species of dwarf mistletoes, Arceuthobium, are especially important in this context in North America, and Phoradendron may also be found on Pinaceae (Hawksworth & Wiens 1996; Mathiasen 2022).
Chemistry, Morphology, etc.. Virjamo and Julkunen-Tiitto (2017) discuss the piperidine alkaloids known from at least some Pinaceae from both subfamilies; there is variation in the synthetic pathway involved, even within a genus like Picea.
The diameter of first order roots seems to vary considerably (W. Chen et al. 2013: rather narrower than co-occuring angiosperms, China, 2016: rather broader than co-occuring angiosperms, U.S.A.). The woolly adelgid Adelgis piceae causes compression wood to form without gravitational stimulus in the plants that it attacks (Timell 1986: vol. 3, chapter 20). Krokene et al. (2008; see also Bann (36) discuss resiniferous tissues in xylem and phloem. Schultz (1990) notes that there are no phloem fibres in Pinaceae. Esteban et al. (2023) provide a well-ilustrated outline of variation in wood anatomy, terms following those proposed by the IAWA Committee (2004). Pinus cuticular wax tubules look almost scalloped (c.f. commelinids!), but this is because the tubules are densely aggregated (Wilhelmi & Barthlott 1997). Dörken and Stützel (2012b) discuss the anatomy of Pinus needles; needles of subgenus Pinus are often described as having two vascular bundles, but in fact there is a single vascular bundle, but in two parts that are separated by a parenchymatous band, the whole being surrounded by a common bundle sheath. Adult plants of Pinus have scale leaves alone on their long shoots; seedlings may bear needles directly on long shoots (see also Dörken et al. 2010; Dörken 2012). For seedling (?hypocotyl) anatomy of Pinus and Larix, see Miller and Johnson (2017); there is potentially interesting variation in features like the presence of cortical sclereids, pith lignification, and number of protoxylem poles.
Dörken and Stützal (2024, see also Dörken & Nimsch 2015; Dörken 2017) suggest that in Pinaceae the pollen cones are pseudanthial... The pollen wall of Pinus is rather low in flavonoids compared with that of the other spermatophytes examined (J.-S. Xue et al. 2023).
The seed coat of Cedrus is vascularized. The seed wing of Pinaceae is derived from the middle or stony layer of the integument. J. Doyle (1963) noted the four-tiered nature of the early embryo; the suspensor tier develops from cells which in Cupressales might well be part of the embryo proper; certainly, they are not positionally homologous with the suspensor cells of Cupressales. Cleavage polyembryony is common, and true polyembryony (more than one archegonium is formed) even more so (Roy Chowdhury 1963), but the seed generally contains only a single embryo. Roy Chowdhury (1963) noted that the sequence of cell divisions of the early embryo of Pseudotsuga taxifolia was somewhat different from that in the rest of the family.
For Pinus, see e.g. Mirov (1967: monograph), Richardson (1998: ecology and biogeography), and Farjon (2005a: monograph); for Pinaceae more broadly, see Page (1990), Gernandt et al. (2011b), Farjon (1990, 2008, 2017) and the Gymnosperm Database, all general. For wood anatomy, see Esteban and de Palacios (2009) and Esteban et al. (2009), both Abietoideae, for leaf anatomy, see Yao and Hu (1982), for epidermal features, see Khan et al. (2019), for details of reproduction, see Roy Chowdhury (1961: Cedrus), Owens and Molder (1979: Picea), for aspects of ovuliferous cone morphology and anatomy, see Hu et al. (1989), Napp-Zinn and Hu (1989), and Gernandt et al. (2011a), for ovules, see Takaso (1984), for the embryo and its development, see Buchholz (1931) and Doyle (1963), for seed coat development, see Owens and Smith (1964).
Phylogeny. Relationships within Pinaceae suggested in the past have depended in part on the kind of data analysed (morphology, molecules) and methods of analysis (parsimony, Bayesian) - see Tsumura et al. (1995), X.-Q. Wang et al. (2000), Rydin and Källersjö (2002), Liston et al. (2006b), and Gernandt et al. (2008, 2016, 2018a, b). For instance, studying the mitochondrial rps3 gene, Ran et al. (2010) found that Larix and Pseudotsuga were sister to all other Pinaceae. However, a problem has been the position of Cedrus with respect to Abietoideae (Abies, Keteleeria, Nothotsuga, Pseudolarix, Tsuga) and Pinoideae (Cathaya, Larix, Picea, Pinus, Pseudotsuga) (Holman et al. 2010). Thus Wang et al. (2000) placed Cedrus sister to the rest of the entire family, Gernandt et al. (2008) and Z.-D. Chen et al. (2016) as sister to Abietoideae (see also C. Hou et al. 2015; Leslie et al. 2018), while Liu et al. (2010) retrieved the clade [Cedrus [Abies + Keteleeria]] as sister to the rest of the family, although Tsuga and Pseudolarix were not sampled; Cathaya and Pinus formed a clade. Holman et al. (2010) nicely summarize the morphological evidence that is compatible with the relationship of Cedrus to either of those groups, or as sister to the whole family; here it is included in Abietoideae (see also Leslie et al. 2018). There was little support for the monophyly of Pseudolariceae or the position ofCathaya as sister to Pinus in Stull et al. (2021).
In the various analyses in Gernandt et al. (2016: inc. morphological analysis with 158 characters, 2018b) the position of Cedrus was uncertain, and the immediate relationships around Pinus the same; fossils - the focus of this study - tended to be unstable in position. Note that fossils assigned to Pityostrobus are scattered through the family phylogeny (Klymiuk & Stockey 2012; Ryberg et al. 2012; Gernandt et al. 2016, 2018b). Gernandt et al. (2016) found that adding morphology to molecular data for extant taxa improved resolution and support, adding fossils to a morphology-only data set for extant taxa decreased support and resolution, when included in joint analyses they reduced support for other relationships in the tree, and using implied weighting (weighting in inverse proportion to homoplasy) improved things - one bottom line is that adding incomplete fossils helps little in phylogenetic analyses.
In a study with exhaustive sampling of conventional Pinaceae and all other Pinales (but not Gnetum, etc.), Leslie et al. (2012, 2018) found the set of relationships [[Cedrus [Pseudolarix [Nothotsuga + Tsuga]] [Abies + Keteleeria]] [[Pseudotsuga + Larix] [Pinus [Cathaya + Picea]]]]. The same two major groups were recovered by Lockwood et al. (2013), although major groupings were not the focus of that study and details of relationships within the two groups differed; see also He et al. (2012), Klimiuk and Stockey (2012), Ruhfel et al. (2014), Y. Lu et al. (2014: Cedrus sister to the first group mentioned above, but other relationships there scrambled, second group o.k.); C. Hou et al. (2015), Sudianto et al. (2016) and Stevens et al. (2016: [[Abies [Pseudotsuga + Larix]] [Pinus + Picea]]).
For the phylogeny of Pinus, see Price et al. (1998), Syring et al. (2005), Gernandt et al. (2005, 2008, 2011a, 2018a: some conflict between plastid and nuclear genes, b), Eckert and Hall (2006); Tsutsui et al. 2009: conflict esp. with mitochondrial genes in subgenus Strobus; Parks et al. (2012) and Gallien et al. (2015). Jin et al. (2021: sampling pretty good) based relationships on an analysis of 1,662 genes from transcriptome sequences, and the relationships they obtained/classification they used are followed here. Pinus has two subgenera (see Jin et al. 2021 for an infrageneric classification), and leaves of subgenus Pinus, the hard pines, apparently have two vascular bundles (but see above), the plesiomorphic condition, while those of subgenus Strobus, the soft pines, have but a single bundle (references in Gallien et al. 2015 for studies on individual subgenera). Lockwood et al. (2013) provide a detailed phylogeny of Picea, sister to Pinus, but Picea was found to be embedded in Pinus by Sen et al. (2016). The recovery of conflicting relationships in this genus may reflect ancient hybridization, mitochondrial introgression and plastome recombination (Ran et al. 2015; Sullivan et al. 2017).
Analysis of nuclear ITS variation was largely uninformative in suggesting relationships between sections in Abies (47 spp.), but at lower levels was more useful (Xiang et al. 2009); in a more extensive study (genes from all three compartments), Xiang et al. (2014) largely resolved relationships in the genus. Z.-R. Wei et al. (2025: RNA-seq, around 1533 orthogroups) looked at 57 taxa in Abies; there probably had been ILS causing some confusion in relationships between the Mesoamerican taxa and gene flow between Old World taxa.
Classification. If the topology suggested by Leslie et al. (2012) holds up, a two subfamily classification, Abietoideae and Pinoideae, the subfamilies being the two major clades recognized there, is reasonable (see above) - however, confirmation of this topology would be useful...
Fot a sectional classification of Abies, with section Balsamea of suspected hybrid origin, see Xiang et al. (2018). Haploxylon/white/soft pines, with (four to) five (to eight) needles = Pinus subgenus Strobus, Diploxylon/yellow/hard pines, with (one to) two to three needles = Pinus subgenus Pinus. Jin et al. (2021) suggested dividing Pinus into these two subgenera, four sections and thirteen subsections (c.f. Gernandt et al. 2005).